Marine Microbes
Introduction
Invisible to the naked eye, there is a teeming world of microbes living in the ocean with a complexity and diversity that rivals all other life on Earth. They include bacteria, viruses, archaea, protists, and fungi. If you weighed all the living organisms in the ocean, 90 percent of that weight would be from microbes. Just because these microbes can’t be seen does not mean they are unimportant. Microbes are often the engines of ecosystems that otherwise would not have access to the food and nutrients they need. Many are also the keepers of healthy ecosystems, cleaning the ocean of waste and often defending against disease rather than spreading it. Microbes live in some of the most extreme environments, from boiling hydrothermal vents to underground glacial lakes in the Antarctic. They were even the first life on the planet, living without oxygen in an ancient ocean. They can glow, help shape clouds, and produce one of the deadliest toxins in the world.
Microbes are essential for a thriving ocean ecosystem. Without them, the world we know would not exist.
Are You An Educator?
What is a Microbe?
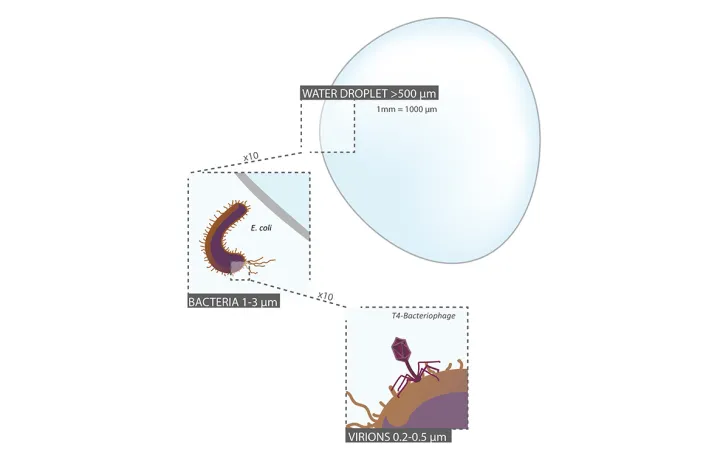
Microbes are everywhere, including the ocean. A single liter of seawater has about one billion bacteria and 10 billion viruses. But what does it mean to be a microbe? A microbe is an extremely tiny organism, and it is not necessarily within a unified group that is closely related. In fact, microbes exist in all three domains of life. There are microbes that are bacteria, microbes that are archaea, and microbes that are part of the more complex domain that plants and animals are a part of—the eukaryotes. Even viruses, a group scientists debate is even a form of life, are considered to be microbes. Microbes are incredibly diverse and live in bustling microscopic worlds that are invisible to our eyes.
You can see how all these microbes relate to one another in our interactive marine microbe tree of life or read on to learn more.
Bacteria
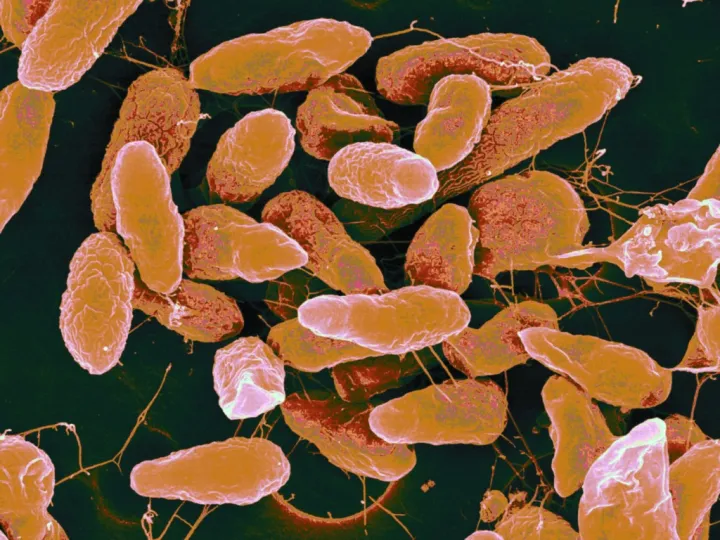
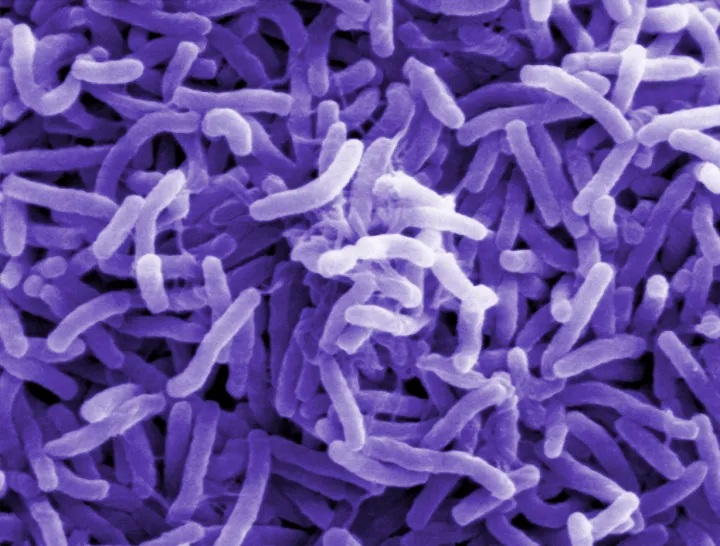
Often regarded as vectors of disease, the majority of bacteria are actually harmless and in fact integral to ecosystems across the globe. They are also extremely abundant—in just a single drop of water there can be over 100 species of bacteria. They have been found in almost every surface on Earth, even in places that are seemingly inhospitable. The smallest are one-hundredth of a millimeter and the largest, found in ocean sediments off the coast of Namibia, are three-quarters of a millimeter, large enough to see with the naked eye. While some obtain energy through photosynthesis, others are able to produce energy through different chemical reactions. Bacteria from the Bdellovibrio genus will even hunt down and consume other bacteria. Some bacteria, like cyanobacteria, lived more than 3.5 billion years ago during the Precambrian. Evidence of their existence can be found in fossilized mounds called stromatolites, seemingly unremarkable rocks which are, in fact, layers upon layers of dead cyanobacteria.
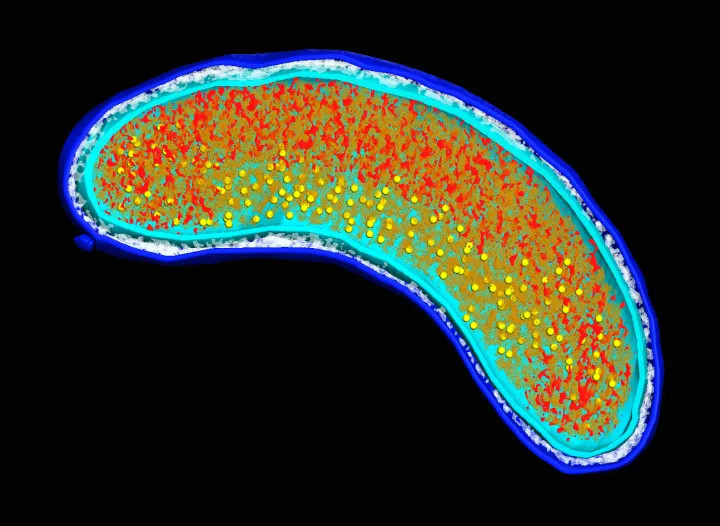
A bacterium is a unicellular organism, or prokaryote, with a relatively simple cell structure. They are usually shaped as either a rod, sphere, or spiral, although some have unique characteristics like a particularly tight spiral coil. The cell consists of a permeable cell membrane, DNA, protein factories called ribosomes, and a protective outer cell wall. Unlike more complex organisms, such as eukaryotes, bacteria lack an enclosed nucleus and instead the DNA floats in a bunched tangle called the nucleoid. Bacterial DNA is usually organized into a single circular chromosome. Bacteria occasionally carry DNA in smaller rings known as plasmids. Often a single plasmid is transmitted from one bacterium to another in a sex-like interaction called conjugation.
Bacteria in the ocean adapt to be best suited for their environment. Those that are close to the water’s surface, like cyanobacteria, tend to be photosynthesizers. It makes sense, then, that the types and numbers of bacteria present at the surface are often tied to location and the season—they take advantage of when the temperature and sunlight are just right. Some bacteria feast on dying phytoplankton and only grow in number when there are seasonal algal blooms.
At deeper depths the bacteria develop unique adaptations to make do without sunlight and, in general, this leads to greater bacterial diversity at depth. About 70 percent of the ocean’s microbes live in the dark open ocean. Here, bacteria are more likely to be efficient swimmers with tails called flagella, a necessity for avoiding hungry predators. Others cling to falling masses of dead particles, the decaying matter a perfect source of nutrients.
Archaea
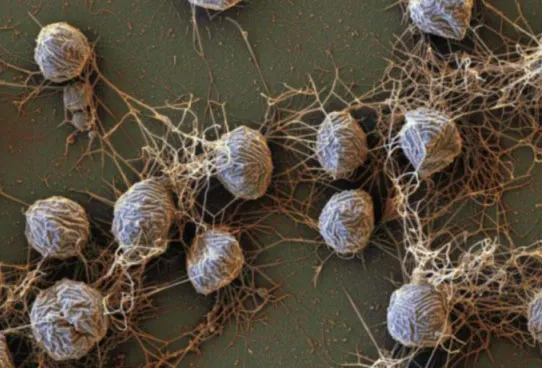
Although once considered specialized bacteria, archaea are now known to be their own unique domain of life. Much of the confusion stems from their visual resemblance to bacteria. They are also single-celled organisms with a basic cell structure. But a closer inspection reveals many of their genes are more similar to complex life, like animals. Many archaea live in the most extreme environments on the planet, including places that are incredibly salty, hot, acidic, or radioactive. Even in the scalding hot springs in Yellowstone, these creatures thrive. They hold the record for the highest tolerated temperature at 266 degrees Fahrenheit (130 degrees Celsius), and some research suggests they could tolerate up to 302 degrees Fahrenheit (150 degrees Celsius). Some archaea can withstand environments close to 35 percent salt—shockingly salty considering ocean water only measures at 3.5 percent.
As the first discovered archaea were found in extreme environments, initially archaea were classified as “extremophiles.” However, it is now known that they live everywhere, in places as dissimilar as the ocean floor to the cow gut. About 40 percent of the microbes living in the ocean are archaea. Some are large enough to see with the naked eye, like one species that grows in white, filamentous mats on mangrove roots in the West Indies, though most are microscopic. Some live associated with corals, sponges, and fishes while others, like Thaumarchaeota, are a dominant presence floating in the water column as tiny plankton.
Viruses
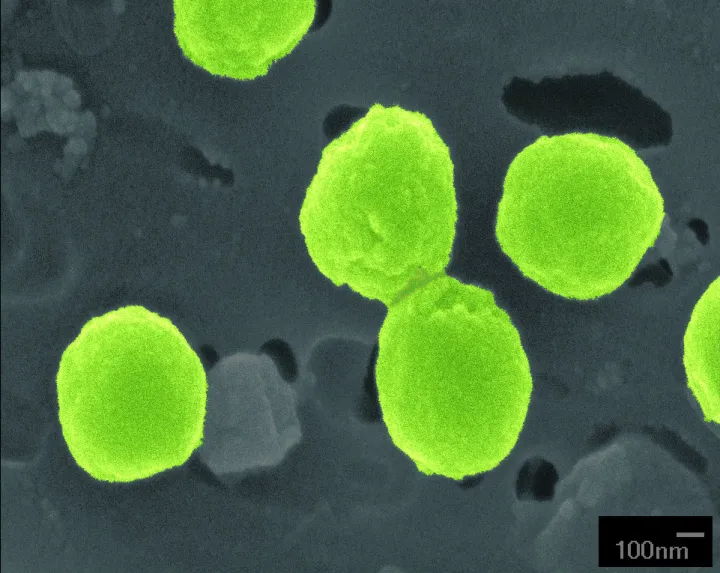
It may seem like an outlandish claim, but we live in a world dominated by viruses. There are more than 1030 viruses in the ocean, a number so high it is about a billion times larger than the number of stars in the known universe.
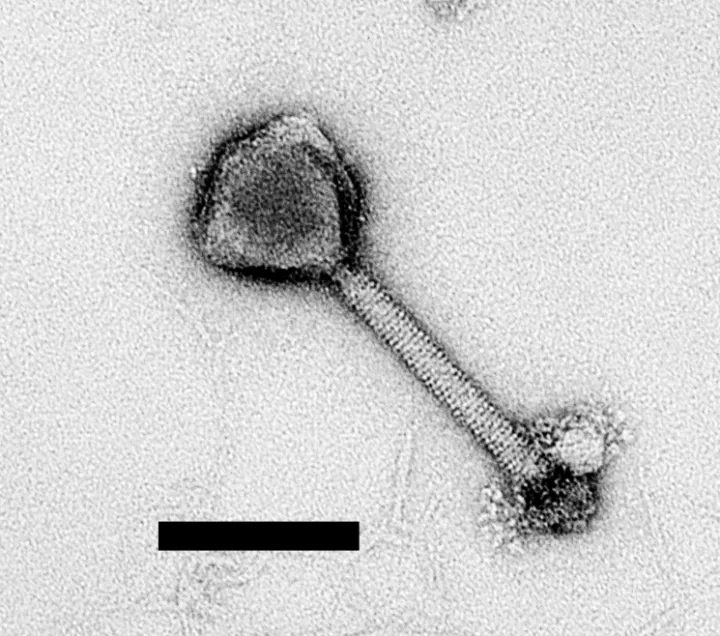
A virus is an infectious agent that must occupy a host cell (like a bacterium) to replicate. Their basic anatomy includes a protein casing called a capsid that holds its genetic material, which is either DNA or RNA. The viruses that infect bacteria and archaea are called phages. Some viruses also have a membrane called an envelope that lines the inside or outside of the capsid. Upon infecting a host, a virus hijacks the reproductive machinery of the cell which then becomes a virus replicating factory. The ability to replicate is often considered a defining characteristic of life so scientists often debate whether viruses can be considered alive.
Viruses that exist in the ocean often infect specific hosts and so when the bacterial communities change, so do the types of viruses present in the ocean. In general, infection rates are higher in the sunlit portion of the water column. Prochlorococcus is a photosynthetic bacteria that is particularly prone to infection, and so when there are high levels of Prochlorococcus in the water, the number of viruses is also high.
Protists
Protists
Some microbes are single-celled, but contain the same inner machinery (called organelles) as more complex multicellular organisms like plants and animals. These organisms are classified as protists and can have similar characteristics of plants, animals, or both. Some get energy from their chloroplasts, the organelles responsible for photosynthesis, and are considered microalgae, while others have whipping tails called flagella that they use to propel themselves through the water in pursuit of bacteria and other, smaller protists that they engulf and ingest. One type of protist, called a choanoflagellate, flushes water through a fringe of 30 to 40 hairs with a flagellum to trap the bacteria, and one dinoflagellate called Pfiesteria piscicida (“fish killer”) uses a feeding tube to suck the fluid from fish tissue, an act that can kill the fish. Others can both photosynthesize and consume prey.
Although small, protists can have dramatic impacts on ecosystems. Many can quickly increase in number to form toxic algae blooms, while others are such voracious predators that they keep the number of bacteria in check. Coral rely on their symbiotic zooxanthellae, a type of protist called a dinoflagellate, to supply them with food and in some tropical areas of the world the water will glow blue-green or red from bioluminescent protists. Despite their importance, protists are some of the least understood microbes in the ocean.
Fungi
Molds, yeasts, mushrooms, and toadstools makeup an incredibly diverse group of fungi on land, but in the ocean, they are comparatively hard to find. Most marine fungi are associated with the decomposition of plant material and can be found in salt marshes, deep sea sediments, and mangrove forests, although they are also found in nearshore mud, animal digestive tracts, deep sea hydrothermal vents, and on both algae and corals. While usually found in shallow water, the fungi Rhodotoula was discovered 11,000 meters below the sea surface in the Mariana Trench. Other fungi live as parasites within marine plants, algae, and animals.
It is challenging to study the extremely small fungi in their preferred marine environments, where nutrients are abundant, but they play important roles in the ecosystem—from recycling nutrients to infecting larger species, such as coral or lobster.
Ecological Relationships
In the Food Web: The Microbial Loop
For much of history, scientists understood the ocean food web to consist of producers, like plants and algae, and consumers, like fish and mammals. Essentially, big things eating little things. But this left out an integral piece of the food web puzzle. Invisible to the naked eye, microbes were not considered important.
But this is far from reality. Microbes contribute to the ocean food web through the microbial loop, a process where microbes recycle the waste of other organisms and loop both the resources and energy remaining in that waste back into the food web. Microbes absorb or “feed” on the microscopic remnants of dead organisms and their waste products, material that would otherwise accumulate and pollute the ocean. When other creatures eat the microbes, they pass on that energy and those resources up the food chain.
The microbial part of the food chain is a world of foreign looking creatures. Bacteria are consumed by creatures called nanoflagellates, tiny orbs with a tail or two and sometimes a skirt. Those nanoflagellates are then engulfed by slightly larger creatures called ciliates, amorphous predators with hairs covering their body. It is only now that organisms large enough to see with the naked eye appear. Copepods, bug-like crustaceans, and other zooplankton feed on the ciliates and are the main food source of small fish.
It is this microbial loop that keeps the ocean clean and clear. Without microbes, the ocean would be a wasteland.
The Scent of the Sea
Sulfur is not only used as a source of energy—it can also be used as a type of chemical defense. By breaking down chemical compounds produced by phytoplankton, bacteria release the aromatic dimethyl sulfide (DMS) into the atmosphere. In small doses DMS produces a subtle aroma, which for many people is the pleasant yet salty and pungent smell of the sea, however, concentrated doses from an algal bloom cause an unsavory scent.
Many algae species are immobile and therefore must heavily fortify themselves against whatever comes their way. If a cold spell should cross where they live they cannot simply get up and leave to find warmer waters. Nor can they leave if a school of voracious predators comes swimming by. Instead they can produce a myriad of protective chemicals that act like bug spray or antifreeze. One particularly popular chemical produced by marine algae is called dimethylsulfoniopropionate. Quite the mouthful, scientists refer to it as DMSP. Although the exact use of this sulfur-rich chemical is still a mystery, scientists guess it can aid in anything from deterring predators, avoiding viral infection, protecting against ultraviolet radiation, regulating salinity and temperature, and possibly acting as an antifreeze.
Algae create a lot of DMSP, as much as 6 x 107 gigatons annually. The majority of algae can’t break down DMSP and when they produce this chemical they also lock away large amounts of sulfur. These algae have created quite the conundrum for themselves— they need the sulfur to build proteins. This is where microbes come into play. Bacteria contain special enzymes that convert the DMSP into usable forms of sulfur. As much as 90 percent of the DMSP produced by algae is converted into consumable sulfur by bacteria. The remaining 10 percent is converted into DMS, an extremely volatile substance that floats into the atmosphere as a gas and is the source of the sea spray smell. A microscopic algae called Phaeocystis is a particularly prolific DMSP producer that can also transform the chemical into DMS. A Phaeocystis bloom can cause a 10 to 100 times increase of atmospheric DMS when compared to the average amount found near the open ocean.
The scent of DMS is a powerful lure for seabirds. Albatrosses, shearwaters, and petrels have a keen sense of smell and they follow the scent of a phytoplankton bloom because it promises tasty krill and fish that feed on the algae. DMS in the atmosphere can also lead to the formation of clouds. For a cloud to form, water droplets must condense onto bits of floating particles, like dust, fire soot, volcanic ash, car exhaust, or DMS. Since cloud formation helps block and repel solar radiation away from Earth back into outer space, the production of DMS by bacteria acts as a potential regulator of Earth’s temperature. A world with more phytoplankton, and therefore more DMS, could have a small cooling effect on the globe.
Symbiotic Relationships
Microbes live in communities and rely on one another to live. Within the community, individual types of microbes can have roles that support the overall well-being of the group. For example, decomposition is a process where microbes are breaking down dead organisms to get at the carbon locked away (see “Microbes as Metabolic Engines” section). This takes multiple steps, and so sometimes microbes will live together and specialize—each individual species will carry out one step of the decomposition process. In another example, microbes may alter the environment to make it a better place to live. One bacterium is able to secrete an acid so that the environment is at the correct acidity (pH) for others to join. Scientists are just scratching the surface when it comes to understanding how microbes interact with one another, and this area of research is sure to reveal some interesting relationships in the near future.
While the majority of symbiotic relationships are between microbes, in some cases microbes will pair up with a larger organism. These microbial relationships make up a microbiome.
The microbiome is a term for the microbial community that takes up residence within the body of another organism. Many of the microbes that live on the surface or within the body of another species play a critical role in the survival of their host, and in return they get a safe place to grow with plenty of food. While for the majority of microbial relationships the exact benefits each symbiont provides remains unclear, often the microbe supplies food, a defense mechanism, or boosts disease immunity. The gutless, bone-eating worm Osedax frankpressi would not exist without the microbes that live within their cells and break down the collagen, cholesterol, and lipids from the skeletons of whales that die and fall to the ocean floor. The Pompeii worm, a feathery worm that lives near hydrothermal vents, can withstand temperatures up to 176 degrees Fahrenheit (80 degrees Celsius) by shielding its tail-end with heat resistant bacteria. Microbes can also act as symbionts for other microbes. In the intertidal, algae and fungi live together and form lichens. The fungi build tough structures that help the lichen stick to rocks despite pounding waves, and the algae supplies food via photosynthesis.
For the blowfish, a familiar delicacy in Asian cuisine, bacterial symbionts supply an important defense against predators—they produce the neurotoxin called tetrodotoxin. For many predators, including humans, it is a deadly toxin. Exposure to the toxin can shut down muscle contractions critical to a beating heart, rendering the toxin a powerful defense against hungry predators. Many other animals besides the blowfish also rely on the toxin to fend off unwanted attackers—blue-ringed octopus, sea stars, horseshoe crabs, the flower egg crab, and several land dwelling creatures use the toxin, which caused scientists to wonder about why so many distantly related species use the same toxin. Current research suggests that symbiotic bacteria are the source of the deadly toxin. Blowfish and other species obtain the bacteria from the environment and sequester the toxin in their organs. Toxic species can tolerate concentrations of the neurotoxin that are 500 to 1,000 times what a normal species could withstand, owing to specialized proteins within their neurons that have evolved to resist the toxin.
Whether as a defense mechanism to avoid predators, an enticing lure to attract prey, or as a means to attract a mate, for many organisms, light plays a critical role in day-to-day life. While some are able to produce light on their own, others acquire the ability to glow with the help of a microbial partner. Anglerfish use tiny glowing bacteria called Photobacterium, which take up residence in the fish’s esca (the “lure”), the structure at the end of its “fishing rod.” Each species of anglerfish pairs with a unique species of luminescent bacteria.
The bobtail squid, too, relies on a luminescent bacterium called Vibrio fischeri, and will selectively allow this bacterium to grow within its light-producing organs called photophores. At birth, a young bobtail squid lacks the bioluminescent bacteria and must find the light-producing microbes in the water column. At this stage of life, the squid’s light organ is not fully developed but small hairs along the photophore sweep the bacteria closer, and a molecular deterrent prohibits all bacteria except Vibrio fischeri from entering. Once one bacterium successfully enters the photophore it multiplies by the hundreds of thousands, a colonization that spurs the full development of the photophore. Without the bacteria, the bobtail squid’s photophore will not develop, rendering the light organ useless as a cloaking device. Vibrio fischeri is a common bioluminescence partner with many other creatures that owe their glowing skills to the microbe.
Health and Disease
A Healthy Checkup
Microbes have a bad reputation as disease causing germs, but the majority of microbes are actually essential for many organisms to survive. These healthy co-inhabitants contribute to the well-being of their partners by providing services the host cannot perform on its own. Some microbes are known to produce anti-microbial molecules that keep out unwanted bacteria, fungi and archaea. Even just the presence of good microbes can be enough to deter unwanted, harmful microbes from colonizing a host’s body, since any newcomer must compete for space and resources. Microbes can also directly influence gene expression within a host’s cell, which changes what the cell produces. Again, current research only glosses the surface when it comes to the microbiomes of marine organisms. At the moment, most studies are concerned with figuring out what microbes exist with the eventual goal to pinpoint the roles of specific microbes and how they impact their host.
One example is a study of whale exhalations from the blowhole that promises to reveal a method of assessing whale health solely on changes in their respiratory microbiome. Scientists are able to use a collection dish attached to a hovering drone to swoop in over a whale just as they are about to exhale and catch the microbe-filled air. Once scientists establish which bacteria should exist within a healthy whale they can then compare this baseline microbiome to other whales to determine if they are ill.
Ocean Outbreaks
Just like in humans, disease is a part of life in the ocean. Although the majority of microbes are benign ocean dwellers, a select few are vectors of disease. Infectious diseases are caused by viruses, bacteria, fungi and protist parasites, which are all referred to as pathogens. Most fish in the ocean are host to a slew of parasites and pathogens despite appearing to be more or less healthy.
But, sometimes, a disease begins to spread, and the outbreak can have significant ecological impacts. Oftentimes, an outbreak is caused by a combination of environmental factors and the presence of a pathogen. Cloudy water, pollution, acidic waters, storms, heat waves, other stressors, or even other pathogens can cause a potential host to become more susceptible to infection. Infected corals combat damage inflicted by parasitic fungi that bore tunnels through their calcium carbonate skeletons by producing a fine, crystalline patch that blocks the fungi. But scientists worry this defense mechanism may become ineffective in acidic oceans. Often disease spread goes unnoticed in the ocean, and it is only when the infected are commercially important or charismatic that people take notice. Some of the more notable marine diseases of the past century include a seagrass wasting disease in the North Atlantic during the 1930s, an ecosystem changing outbreak of white band disease in Caribbean corals during the 1980s, a white spot disease outbreak in shrimp during the 1990s, and lobster shell disease from the late 1990s to the early 2000s.
Between 2013 and 2015 marine diseases were put in the spotlight when a mysterious pathogen began infecting sea star populations up and down the coast of the western United States. In total about 20 different species were infected by the sea star wasting syndrome, an illness that effectively turned sea stars into a soupy goo. While some sea stars species were able to fight off infection, others like the sunflower star (Pycnopodia) would completely disintegrate only a week after the first signs of exposure. In Vancouver, more than 90 percent of the sunflower star population was killed by the disease. Not all sea star species begin to show signs of the illness when exposed, suggesting there may be multiple pathogens and environmental conditions that trigger the syndrome.
A Warming World
Rising ocean temperatures provide the perfect environment for the spread of disease where generally, most bacteria, viruses, and fungi can thrive in warmer temperatures. Temperature acts like a fast-forward mechanism within the microbe’s cell, speeding up chemical reactions and increasing mutations at the genetic level that can then lead to new strains. Microbes are also able to expand their habitat as they follow warming waters and altered ocean currents where they encounter new hosts. From 1990 to 1991, warmer than normal water caused an infectious oyster parasite to spread 500 km (310 miles) north from the Chesapeake Bay up to Cape Cod and eventually appear off the coast of Maine a few years later. Outbreaks of Vibrio, a genus of bacteria that infects oysters (and causes cholera in humans), now occur more frequently in northern latitudes. For the first time, Vibrio made its way to Alaskan waters during a particularly warm summer in 2004.
Not only have new hosts never before been exposed to these encroaching pathogens, they themselves are battling with the stress of a warmer climate and are therefore more susceptible to infection. Many of the microbial infections that plague corals only appear after the coral has become stressed enough by a severe storm or heat-related bleaching. In 2005, corals in the Caribbean Virgin Islands began recovering from a severe bleaching event only to succumb to white plague disease, an outbreak that killed 50 percent of corals in the region.
Habitats
From deep within the earth’s crust to lakes under glaciers in the Antarctic, microbes are colonizers of some of the most extreme habitats on the planet. They can withstand crushing pressures, temperatures hot enough to melt lead, and thousands of years on the edge of starvation. Even the surfaces and guts of marine creatures are filled with microbes, often unbeknownst to their host.
Water Column
Living in the water column near the ocean surface and sunlight is a great place for a microbe to thrive. This area is home to some of the world’s biggest photosynthesizers.
The most impressive is a cyanobacterium called Prochlorococcus. It is estimated to be more abundant than any other photosynthesizer on the planet, and to be responsible for producing 20 percent of the oxygen in the atmosphere. One in every five breaths you take, you owe to Prochlorococcus. Perhaps as amazing is the fact that scientists only discovered this super-abundant photosynthesizer in 1988!
The water column is also home to what is potentially the most abundant microbe on the planet, a bacterium called Pelagibacter. This microbe usually accounts for about 25 percent of all the microbes in the water column, but during a bloom it likely will account for up to 50 percent. Unlike the cyanobacteria, Pelagibacter feeds on dead organic matter that is dissolved in the ocean water, a feeding process that is part of the microbial loop. Microbes like Pelagibacter that live in the water column and feed on dead organic matter help keep the ocean clean and clear.
Seafloor
A sandy ocean seafloor is a great place for microbes to live. The seafloor is about 10,000 times more densely populated than the open ocean, and 99 percent of those seafloor dwelling microbes live on sediment. The bacteria tuck themselves in the protected grooves and divots of the sand grains. A single grain of sand can harbor as many as 100,000 bacteria.
Continue down beneath the seafloor to a world almost completely void of nutrients and light and you will still find microbes. These bacteria are hearty survivors that were buried alive hundreds to thousands of years ago by algae falling from surface waters. As time progresses the bacteria are further buried underground as algae accumulates and the food they subsist on continues to deplete. Starved of nutrients, the bacteria become metabolically sluggish and slow to reproduce. While some bacteria in the water column can double in less than a day, deep underground the same duplication can take hundreds of years. One study determined that bacteria living in a 4,900-year-old sediment layer replicated once every 100 years. But that depth only scratches the surface. Scientists discovered bacteria a mile and half below the seafloor in 20 million-year-old coal deposits, the deepest bacteria that have been found to date.
Biofilms
A biofilm is an interconnected city of microbes including bacteria, algae, protists, and viruses, that forms a film on a hard surface, like a rock or seabed. As unicellular organisms, microbes often float alone or in small colonies in the open water, but if they drift close to a surface like a rock or seaweed they can attach and become stuck to the surface. Once the first microbe gets caught, a domino effect ensues where microbes will continue to cling onto one another almost as though caught by velcro. As the microbes pile up they can form layers of unique microbe communities which can eventually build into microbial mats several millimeters thick. Once a microbe attaches to a biofilm it will undergo a series of changes so that it will be best suited for an immobile existence surrounded by a network of other microbes. The most important change is the production of sticky cement secretions that glue the microbes together. A 2008 study found that biofilm bacteria produced a toxin that deterred grazing by a bacteria-eating protist, suggesting that life in a biofilm has several advantages when compared to floating exposed in the open ocean. Vibrio cholerae, the bacteria responsible for cholera, naturally lives as a biofilm covering zooplankton, a discovery made by Rita Colwell.
Hydrothermal Vents
Deep beneath the ocean’s surface, hydrothermal vents are hotbeds for life. At first inspection, it seems unlikely—from cracks in the earth’s crust spew scalding water that has been heated deep within the earth to temperatures up to 752 degrees Fahrenheit (400 degrees Celsius), a temperature hot enough to melt lead. These vents are also so deep that they never see a glimmer of light from the sun. Despite these obstacles, clams, mussels, shrimp, and gigantic worms thrive in these habitats. Their existence is thanks to bacteria.
Life by a hydrothermal vent relies on the energy produced by symbiotic bacteria. The bacteria live either inside the bodies or on the surface of their hosts. But unlike most life on Earth that uses light from the sun as a source of energy, these bacteria produce energy through a chemical reaction that uses minerals from the vents.
Scientists first learned of these symbiotic relationships through the study of the Riftia tubeworm. Upon first discovering hydrothermal communities in 1977, scientists were perplexed by the diversity and abundance of life. The worm’s blood red plumes filter the water and absorb both oxygen and hydrogen sulfide from the vents. Hydrogen sulfide is normally toxic, but the Riftia worm has a special adaptation that isolates it from the rest of the body. Their blood contains hemoglobin that binds tightly to both oxygen and hydrogen sulfide. Further investigation into these unique habitats showed that many of the other creatures that live by the vents also rely on symbiotic bacteria. The yeti crab waves its arms in the water to help cultivate bacteria on tiny arm hairs which it then consumes.
Coral Reefs
Imagine a coral reef and a vision of colorful corals, crabs, and swarms of fish comes to mind, but coral reefs are also hotbeds for marine microbes. Corals and certain bacteria and archaea form symbiotic relationships in which they rely on one another to live. These microbes can be found covering the outer surface of the corals, deep within their tissue, or among the sediments beneath the reef where there are about 10,000 times the number of bacteria when compared to the surrounding water. Corals will even secrete a protective mucus that provides nutrients for selective “good” bacteria as a way to influence their microbial communities. The microbes in return provide essential services for the corals like supplying nitrogen, breaking down toxic waste, and feeding nutrients to a coral’s symbiotic algae partner, the zooxanthellae (also a microbe). They may also deter the growth of harmful microbes by taking up the livable surface and blocking unwanted settlers or through the production of antibiotics. One study suggests that specific microbes may help corals combat the stress of warmer water, which will become critical to coral survival as intense heat waves increase due to climate change.
Microbes as Metabolic Engines
The majority of life on earth relies on energy obtained from the sun’s rays, either directly through photosynthesis or by consuming a photosynthetic organism and its stored energy. Photosynthesis is a series of chemical reactions that takes the energy from the sun and converts it into a form of energy that cells can use. While many microbes partake in this method for acquiring energy, some live in environments where sunlight cannot reach. In the depths of the ocean, light only penetrates down to 650 feet (200 meters) and some microbes live deep within the sediments. Yet even in the deepest and darkest areas of the ocean they are able to survive. Most microbes subsist off of falling bits of organic material (decaying bodies, food scraps, waste) that come from surface waters, but select others have developed novel ways to produce energy. Instead of sunlight, these specialized organisms obtain energy through chemical reactions, a process called chemosynthesis. All chemosynthetic microbes use chemical reactions to obtain energy, but the chemicals they use differ from species to species based upon the location where they live and what chemicals are available.
Power from the Sun
Carbon is one of the main building blocks of life. All organisms need it to build molecules in their body, including humans. But carbon molecules come in many forms and only certain ones can be used by living things. Carbon dioxide, for example, must be transformed by an organism in order to be used as a building block within a living body. This is often done through photosynthesis, a process that also releases oxygen, but microbes can also accomplish this feat through other chemical reactions. Microbes are amazing carbon transformers and play a critical role in shuttling carbon within the ocean food web. By taking up carbon dioxide from the atmosphere, marine microbes also play a critical role in regulating the earth's climate.
Not all carbon is the same. In fact, the majority of carbon in the environment is locked away in unusable forms. For example, for most organisms, the carbon in carbon dioxide cannot be processed. Marine microbes play an important role in taking carbon dioxide from the atmosphere and transforming it into new carbon molecules called carbohydrates, often known as “carbs”. These carb molecules can then be used as building blocks within the body. This process occurs during photosynthesis—the energy from the sun powers the carbon transformation. Cyanobacteria in the ocean, such as Procholorococcus and Synechococcus, are responsible for almost half of the photosynthesis on Earth! Organisms that can’t photosynthesize obtain their carbon when they eat a photosynthesizer or other creatures that ate photosynthesizers.
Recycling Machines
The majority of microbes, however, tap into a source of carbon readily within reach rather than produce their own.
Invisible to the naked eye, molecular pieces of proteins, lipids, carbohydrates, and nucleic acids (DNA and RNA) drift throughout the water column. Collectively referred to as dissolved organic material (DOM), these molecules are too small for the average consumer to consume but are perfect sources of fuel for bacteria. By consuming the molecules, bacteria are reintroducing critical energy back into the food web when slightly larger creatures, like krill, eat the bacteria.
How do the dissolved molecules exist in the first place? Contrary to what is often illustrated in simple food webs, a good chunk of a meal is wasted by the consumer. Ocean creatures are very inefficient when it comes to eating. Tiny zooplankton chew through phytoplankton and lose much of what they aim to eat. Instead of being consumed, some of the phytoplankton are ripped apart by munching jaws and the cellular innards are released into the open ocean where they become suspended in the water column as DOM. Scientists refer to this process as “sloppy feeding.”
In another process, single-celled protists, like the paramecium, contribute to dissolved organic material when they excrete their waste. After engulfing a phytoplankton in a digestive sack called a food vacuole, the paramecium will use all the nutrients, leaving waste and digestive proteins behind. Once the nutrients have been absorbed, the vacuole fuses with the outer membrane and expels the leftover contents. About 25 percent of ingested carbon is eventually expelled as waste. As the saying goes, “one [wo]man’s trash is another [wo]man’s treasure,” and so the bacteria use what the paramecium has deemed unusable as a source of fuel.
But the largest source of DOM comes from the death of phytoplankton cells due to viruses. The majority of marine viruses operate through the lytic cycle, a process that causes the host cell to lyse, or burst. Once a virus infects a host phytoplankton cell it replicates itself using the cell’s genetic machinery. The host cell then dies, expelling its inner contents, including the newly grown viruses, into the ocean. Every day, about 20 to 40 percent of marine microbes are infected by viruses.
Not only do microbes get a nutritious feast when they eat DOM, they are also serving a critical role in the environment. Microbes clean up this ocean waste, and if it weren’t for microbes the ocean would be mostly full of decay. The microbial loop is so important, scientists view it as the microbes’ most essential ecological role.
Transformational Power
Instead of sunlight, specialized organisms can obtain energy through chemical reactions, a process called chemosynthesis. All chemosynthetic microbes use chemical reactions to obtain energy, but the chemicals they use differ from species to species based upon the location where they live and what chemicals are available.
Carbon
The ripe smell of dead and decaying matter is the stuff of life for some archaea. The production of odorous methane, a carbon-based molecule, by these microbes is a last-ditch effort to use the remaining bits of decaying matter that no other microbes can use to produce energy. These microbes are methanogenic archaea, and they have devised a way to drive energy production with the less-desired leftover carbon-based molecules. These unique archaea chemically transform hydrogen and carbon dioxide into methane to produce energy in a special process called methanogenesis. Methane production by microbes mostly occurs in swampy, waterlogged wetlands, but can happen anywhere oxygen is limited and there is plenty of decaying matter to pull carbon from.
One such hydrogen-rich environment includes hydrothermal vents. Methanopyrus kandleri and Methanocaldococcus are heat-loving species of Archaea that make their home by hydrothermal vents where they have ample access to hydrogen gas, which they convert to methane. Methanocaldococcus was first discovered at the base of a white smoker in the middle of the Pacific Ocean while Methanopyrus kandleri has been isolated from hydrothermal sediments at Kolbeinsey Ridge off the coast of Iceland and the Guaymas Basin in the Gulf of California. In the laboratory Methanopyrus kandleri cells can even divide at 122°C, the highest temperature known to be compatible with microbial growth, though it grows best at 98°C. Methanopyrus translates to “methane fire.”
While for some microbes, methane is a waste product, for others methane is their source of energy. Both archaea and bacteria are known to break down methane in the process of producing energy. About a half mile beneath the glaciers of Antarctica lies a lake that is teeming with microbes. A subset of these microbes are methanogens that live in the lakebed soil and feed off of the decaying plant and animals that were trapped in sediments millions of years ago. Surprisingly, there is very little methane in the lake water. That’s because a methane-eating bacteria is consuming more than 99 percent of the methane produced in the lake sediments.
Microbial fungi can also play an important role in breaking down complex carbon molecules that are locked away in dead plant and animal carcasses. The fungi secrete special compounds into the environment that then break apart the complex structures. Often, carbon dioxide is released, providing other marine organisms with an essential resource. In deep seafloor environments, fungi can be a dominant presence in the microbial community and play an essential role in recycling carbon.
Sulfur
For many organisms, including humans, oxygen is required in the production of energy. That is why we breathe in oxygen. But in places where there is no oxygen, microbes have a way to make ends meet—they turn to sulfur molecules. The molecule sulfate fills the same role as oxygen in the production of energy.
The use of sulfur is particularly important near hydrothermal vents, deep ecosystems lacking any sunlight, which often spew hydrogen sulfide into the surrounding ocean water. The superheated “black smoker” vents that get their color from various minerals provide microbes a source of hydrogen sulfide, a molecule that—when paired with oxygen—releases energy. Entire ecosystems that include worms, crabs, and fish rely on the ability of these microbes to produce energy from the sulfur compounds. Other microbes near vents get their energy from hydrogen gas and produce hydrogen sulfide from sulfur compounds spewing from the vents. At a unique system near the West Mata volcano off the coast of Samoa it is believed that the mats of microbes that cover the ocean floor around the volcano get their energy from yet another form of sulfur called white sulfate.
Nitrogen
About 78 percent of the atmosphere is nitrogen gas. And, yet, for most living things that require nitrogen to survive this is a problem. Gaseous nitrogen is impossible for most phytoplankton to process. It’s the same concept as a thirsty, marooned sailor surrounded by an ocean of seawater—water, water everywhere but not a drop to drink. Just like humans require freshwater to survive, the majority of phytoplankton require nitrogen in the form of chemical ammonium (NH4+) or nitrate (NO3-). Bacteria are able to transform gaseous nitrogen into usable forms through a series of chemical reactions. The process of turning gaseous nitrogen from the air to ammonium is called nitrogen fixation.
Bacteria and archaea are also critical for the recycling of nitrogen from dead organisms. When organisms like fish or algae die, the nitrogen within their bodies is locked away in the form of proteins. Special bacteria and archaea take the nitrogen from the protein and transform it into ammonium. Some microbes will take it several steps further to create nitrate, a process called nitrification. Since most bacteria and phytoplankton prefer ammonium and nitrate, the nitrogen stored in the proteins must go through these transformation processes before it can be reintegrated into the food web.
But some bacteria and archaea are able to directly convert ammonium into nitrogen gas. These are the same microbes found in wastewater treatment plants. Often, oxygen is a key component for energy production, (humans and other animals breath in oxygen for its use in cellular respiration) but in a world void of oxygen, these bacteria turn to alternative materials. In 1999, scientists discovered a bacterial process called anammox (short for anaerobic ammonium oxidation) where the bacteria were breaking down ammonium to produce energy. But during this conversion process, the microbes produce a nasty chemical also found in rocket fuel called hydrazine that they then must lock away in special, fortified compartments. Bacteria that rely on anammox are also extremely slow growers, only doubling every 14 days. Cyanobacteria that gain their energy from the sun can double in a single day, and bacteria in a human’s gut double every 20 minutes!
For other microbes who live in environments with limited oxygen, nitrate (NO3-) is used in place of oxygen to produce energy. This is a process called denitrification. During the breakdown of nitrate the microbes produce nitrogen gases including nitrous oxide (N2O), nitric oxide (NO), and pure nitrogen gas (N2), which then float up into the atmosphere. The newly produced nitrogen gas is now recycled and readily available for nitrogen fixating microbes to use for their own energy production.
Limiting Nutrients
In order for microbes to grow they need several key nutrients, including iron. Iron is a critical mineral used in photosynthesis, respiration, and nitrogen fixation. While on land iron is one of the most abundant elements, in the ocean it is rarely found. Whatever iron makes its way to the ocean originally had to come from land—the majority comes from dust particles blown out over the ocean by strong winds or as sediment swept out by rivers. With little iron around, microbes have evolved to become efficient iron hoarders, and secrete special molecules that grab onto passing iron particles. Microbial growth, therefore, is often limited by the amount of iron in the ocean.
Phosphorous is another limiting nutrient that organisms need to live. It is a critical component of DNA and many proteins. But in the ocean phosphorous is often scarce. Like iron, the main source of phosphorous comes from land. In the Sargasso Sea, many microbes in the water column have evolved ways to limit their phosphorous use. Some have adapted to use sulfur instead of phosphorous to build proteins, reserving the phosphorous for building DNA. It is also common in areas of the ocean with low amounts of phosphorous for microbes to have small genomes—if the key building block for DNA is scarce then it is beneficial to have little DNA. Microbes have also developed ways to become efficient phosphorous consumers and make special molecules that are especially good at grabbing phosphorous.
Microbial Research
The Foundation of a New Field
In 1675, Dutch lensmaker Antonie van Leeuwenhoek peered into his powerful microscope and made a startling discovery. The rainwater under his lens held innumerable creatures that “turn themselves about with that swiftness as we see a top turn round.” These “wee animalcules,” as he would call them, were the first observed bacteria. Ecstatic, Leeuwenhoek sampled water from every conceivable location, including the sea. All were home to animalcules.
It wasn’t until over 200 years later though that research into oceanic bacteria began in earnest.
Inspired by the work of Louis Pasteur and his claim that “germs” were found everywhere, Adolphe-Adrien Certes set out to see if that were the case for the deep sea. Using samples obtained during the French Talisman and Travailleur expeditions of the 1880s, Certes was able to grow microbes from samples obtained at depths between 927 to 5100 meters. It appeared microbes were indeed, found just about everywhere.
Dr. Bernhard Fischer was another microbiologist who transitioned from the world of medicine to the world of oceanography. Trained as a researcher of hygiene in Germany, Fischer is best known for his discovery of luminescent bacteria, one of which is now named after him. His marine studies were published in one of the first books about marine microbiology called Die Bakterien des Meeres (Bacteria of the Sea). Despite a handful of microbiologists that delved into the marine world, marine microbiology was still in its infancy as a unique area of study.
By the 1930s and 1940s, marine microbiology finally became recognized as its own field. Claude ZoBell, an American microbiologist, began teaching microbiology at the Scripps Institution of Oceanography and later published the classic textbook Marine Microbiology: A Monograph on Hydrobacteriology. In addition to discovering over 68 species of oceanic bacteria, he pioneered the study of marine microbes and their everyday activities. Because of these efforts, ZoBell is regarded as the “father of marine microbiology.” At the same time just up the coast, C.B. van Niel was influencing students at Stanford University’s Hopkins Marine Station. He would also bring to the United States a Dutch technique that allows for growth of a specific microorganism. This contribution helped reveal the immense diversity of bacteria in ocean environments.
But one of the most influential events in the study of marine microbes during the 1900s was a complete accident. In 1968, Alvin, a Woods Hole Oceanographic Institution submarine, flooded and sank to the bottom of the ocean. Upon retrieval, the crew noticed an interesting item left behind in the sub—someone’s lunch. Despite the 10 months that had passed between when Alvin sank and was retrieved, the sandwich and apple appeared as if they had just been prepared. The discovery of the lunch’s preservation inspired many scientists to question why that was the case and, ultimately, led to increased interest in the role of microbes and decomposition in the deep sea. Many scientists saw this as proof that microbes were not very active in the deep sea, an idea that has now been proven false.
Part of the reason why it is so difficult to study deep sea microbes is because of pressure. At a depth of 6,500 feet the pressure is almost 200 times what we feel on land. In the Late 1970s a concerted effort across three laboratories, those of Holger Jannasch, Aristides Yayanos and Rita Colwell, began to recover uniquely pressure-adapted ('barophilic') microbes from the deep sea. The work eventually led to the discovery of the first microbe to thrive in high pressure in 1979. Later, a microbe that required high pressure to survive was discovered by Yayanos. Colwell and her then graduate student, Jody Deming, used the microbes found in the guts of deep-sea animals to demonstrate that entire communities of microbes can thrive in the deep sea.
The Molecular Era
By the late 1900s, studying marine microbes would require more than simply growing them in the lab and examining them under a microscope. New scientific techniques would need to be developed to reveal more about the microbial world.
In 1976, Carl Woese and George Fox revolutionized the field of microbiology with their discovery of a third domain of life—the archaea. By comparing RNA gene sequences in a revolutionary technique pioneered by the pair, they were able to show that not all microbes were closely related.
Key to Woese and Fox’s discovery was the use of RNA, a type of genetic code similar to DNA. Though eukaryotes, archaea, and bacteria have different cellular structures, they share a common piece of machinery within their cells called the ribosome. The ribosome is the protein factory of the cell, and it builds proteins by reading coded directions based upon the DNA. Ribosomes themselves are built out of RNA molecules, and it is the sequences of this special ribosome RNA that Woese and Fox compared between archaea, eukaryotes, and bacteria. It was also possible to discover new species of microbes using this technique. After mapping out the ribosome’s RNA code scientists could then compare the code to other known microbes. If the code was unique, it indicated the discovery of a new microbe.
Soon other genetic techniques would arise.
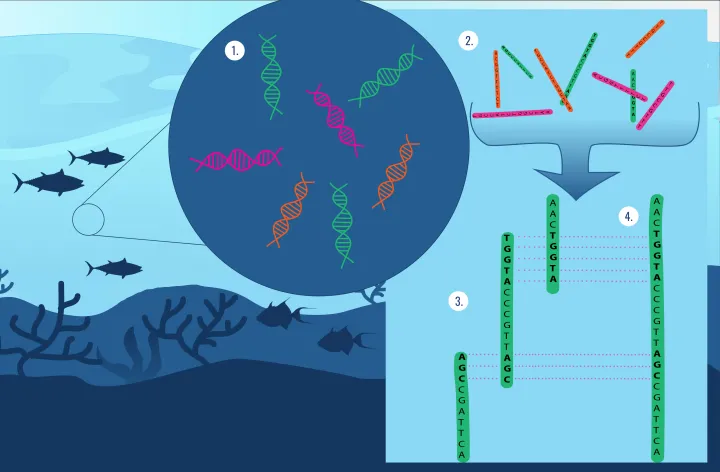
By 2003 and 2004, the first full genome sequences of marine microbes were published. This meant that scientists now knew the exact DNA code for a specific species. The genomes of Prochlorococcus and Synechococcus, two of the most abundant photosynthesizers on the planet, were two of the first microbes sequenced. But isolating a specific species of microbe is not always possible. Many microbes will not grow in the saltwater solutions scientists use in the lab. This poses a problem with many molecular techniques that require a pure sample of only one microbe. The invention of metagenomics would change that.
Metagenomics is the process of taking DNA from the environment and reading the genetic codes to then reveal which species live at that environmental location. It can also tell scientists what kind of microbe is in that location and what it can do. For example, the discovery of photosynthesis genes in the environment tells scientists that a photosynthesizer is present. The discovery of genes that produce methane tells scientists that the microbes in that specific environment are methanogens, microbes that make energy through chemical reactions.
The application of metagenomics to the field of marine microbiology would reveal a whole new world of microbes, including the diversity of marine viruses. This field exploded in 2004 when a technique known as shotgun sequencing, first used to sequence the human genome, was applied in the Sargasso Sea, an area in the middle of the Atlantic Ocean. This has since expanded to countless unique environments across the ocean including a whale carcass on the seafloor.
Today, scientists continue to gain an understanding not only about what microbes exist in the ocean but what they do. Over and over again it is realized that though microbes are invisible to the naked eye, they are integral to and abundant in the ocean world.
You can explore more using our history of marine microbes timeline.
Human Connections
In the Home
You may not realize it, but on a day-to-day basis humans rely on marine microbes, even within the home. As inhabitants of extreme environments, microbes produce specialized proteins and molecules to help them survive harsh conditions. Some of these substances can be applied for human use and are now found in the cosmetic, textile, manufacturing, and fuel industry. The degrading enzymes produced by heat tolerant microbes are perfect stain removing additives to detergents, as they can withstand both the intense heat of the water and the harsh nature of bleach. Microbial enzymes are also heavily used in food processing, both for preservation and to alter the texture, taste or quality of the food. They are often used to tenderize meat, in baking, and to remove lactose from milk for those who are lactose intolerant.
Marine microbes may even have a home in the medicine cabinet one day. Historically, many of our most successful drugs are from terrestrial microbes, and now researchers are turning their search to the ocean. Only seven marine-based drugs are FDA approved, mostly from sponges and mollusks, but scientists are hopeful that this number will multiply as the study and discovery of new marine microbes continues. It is only in the last decade, that over 200 new compounds produced by marine microbes are discovered annually. Now we know that many of the compounds once attributed to sponges are actually from their microbial symbionts. Marine microbes show great promise in yielding tumor-suppressing drugs, Alzheimer treatments, pain alleviating drugs, and antibiotics. One marine bacterium called Serinicoccus produces a compound that causes melanoma cells to implode within three hours. Another is able to stop the growth of both anthrax and the antibiotic-resistant staph bacterium (MRSA), an exciting find considering the recent rise in antibiotic-resistant bacteria. Microbes can also help boost current antibiotics by shutting down harmful bacteria’s defense mechanisms. A marine microbe called Pseudoalteromonas piscicida does this by clogging the machinery that pumps antibiotics out of resistant strains of bacteria.
Dead Zones
Every spring large rivers flush nutrients from far inland out into the ocean. Filled with fertilizer and livestock excrement, it is the perfect concoction of nitrogen and phosphorous for algae to grow. The combination of plentiful nutrients from the runoff and sunshine cause large algal blooms, some of which can be seen from space. Some algae produce toxic chemicals that harm other organisms, but there is another side effect of these harmful algal blooms (HABs)—dead zones. A dead zone is a large swath of ocean that has limited oxygen (a condition called hypoxia) and can no longer support the oxygen needs of animals that breath underwater via gills. As a result, areas once teeming with life become underwater ghost towns. Although dead zones are affiliated with algal blooms, they are actually the result of an explosion of microbial growth. As the algae die they provide a feast for microbes, who multiply and consume oxygen as they help decompose the dead algae. The second largest dead zone in the world occurs annually at the mouth of the Mississippi River in the Gulf of Mexico. Year to year it can vary from under 1,931 square miles (5,000 square km) up to 8,494 square miles (22,000 square miles), or the size of Massachusetts. Seven of the world’s 10 largest marine dead zones are in the Baltic Sea, a statistic that has led the European Union to focus on pollution reduction in the region.
While hypoxia is a natural phenomenon in some regions of the ocean, the influence of human activity in other areas is a cause for concern. Increased pollution over the last 50 years has contributed to an increase in hypoxic areas and dead zones. Warming oceans due to climate change also expand dead zones since an increase in temperature decreases the amount of oxygen that can dissolve into water. In 2019 there are 415 documented dead zones worldwide.
Oil Spills
As a world dependent upon oil as our main source of energy, sometimes oil accidentally finds its way into the environment. Tanker spills, residue expelled from ship ballast water, offshore blowouts, and run-off from the shore can cause oil to accumulate in the ocean and harm marine ecosystems. Fortunately, certain marine microbes are natural oil consumers. They exist, in part, because along the bottom of the ocean there are natural seeps where oil bubbles out from cracks in the seabed. Here, microbes have evolved over millions of years the ability to consume and degrade oil. There are hundreds of species of bacteria, archaea, and fungi that rely on oil and in normal oceanic conditions, these microbes account for less than 1 percent of the microbial community. But in the presence of oil they multiply, eventually becoming 10 percent of the community.
The role of these microscopic oil consumers became critical in the aftermath of the Gulf of Mexico Deepwater Horizon oil spill, recognized as the worst marine oil spill in U.S. history. Because this spill occurred on the seafloor, where oil-consuming microbes were already present cleanup workers turned to dispersants to help aid the tiny microbes.
But dispersants aren’t always an option for oil cleanups. During the second largest oil spill in U.S. water—the Exxon Valdez spill off the coast of Alaska in 1989—high winds contributed to the decision to not use dispersants. Despite efforts to physically contain the spill offshore, about 15 percent of the Gulf of Alaska coastline became oiled. Cleanup teams then turned to bioremediation, or the addition of nutrients to stimulate the naturally occurring microbes to degrade the oil more quickly. Nitrogen fertilizer was added to the coastal waters, a decision that increased the degradation of oil by microbes from three to five times in some areas. Despite being lauded as an overall success, trace amounts of oil remain trapped under rocks and under sediment in the area. Bioremediation is most successful in areas of small pebbles and coarse sand since the added nutrients struggle to break into the tight-knit barrier imposed by fine sand. Other successful uses of bioremediation include the Prestige spill near Spain in 2002 and a spill off the coast of Kuwait during the Gulf War of 1991.
At the Smithsonian
Once overlooked, microbes are now a hot research subject for scientists who have realized that despite their small size, they are immensely important to the existence of healthy ecosystems.
Coral reefs are one such ecosystem impacted by invisible microbial neighbors in several different ways. Scientists at the Smithsonian Marine Station at Fort Pierce are looking at the microbiomes of corals to determine how the presence or absence of certain microbes affect the host coral’s health. Max Teplitski and Valerie Paul are specifically interested in comparing the microbiomes of healthy corals and those that have black band disease, an infection characterized by a mat of diverse microbes that spread across the coral in a visible black band. It appears that healthy corals have a different microbial community than infected corals and now the researchers aim to figure out why that might be.
For many animals that spend the first part of their lives drifting in the open ocean, the smell of microbes is a strong cue to settle down. Several scientists at the Smithsonian are looking into both how the microbes cue the larvae and why the smelliness is a draw for young drifters. Research by Jennifer Sneed found that mustard-hill coral larvae drifting in the open ocean choose their home by following the scent of bacteria. The bacteria in the slimy biofilms that cover ocean surfaces produce “smelly” chemicals with certain species producing different scents. A coral larva can distinguish the smell of a specific species of bacteria and will choose to settle on surfaces that include these bacteria. It is still unclear why the larvae like the smell of one microbe over the other, but Sneed and others suspect it may be related to their chance of survival.
Symbiotic microbes can also have an influence on their host’s genetic evolution. The mollusk Solemya velum, a bivalve like clams and scallops, lives alongside a microbe that produces energy using sulfur-based molecules called sulfite. This mollusk obtains its microbial partners both from its parents, a process called vertical transmission, and from its environment, called horizontal transmission. At the Smithsonian National Museum of Natural History, Vanessa Gonzalez is mapping the genome of the mollusk in an effort to better understand how these two species coevolve.